oil temperature OPEL 1900 1973 Owner's Manual
[x] Cancel search | Manufacturer: OPEL, Model Year: 1973, Model line: 1900, Model: OPEL 1900 1973Pages: 625, PDF Size: 17.22 MB
Page 442 of 625

DIAGNOSIS
SEQUENCE
1. Check and correct oil level. Refer to Specifications
for checking and refill procedures.
2. Check and correct vacuum line and fittings.
3. Check and correct manual linkage.
4. Road test car using all selective ranges, noting
when discrepancies in operation occur.
5. If engine performances indicates an engine tuneup
is required, this should be performed before road
testing is completed or transmission correction at-
tempted. Poor engine performance can result in
rough shifting or other malfunctions.
CHECKING PROCEDURESBefore diagnosis of any transmission complaint is
attempted, there must be an understanding of oil
checking procedure and what appearance the oil
should have. Many times a transmission malfunction
can be traced to low oil level, improper reading of
dipstick, or oil appearances; therefore, a careful anal-
ysis of the condition of oil and the level may elimi-
nate needless repairs.
When checking oil level in the Opel Three Speed
Automatic Transmission, the procedure outlined in
Specifications should be followed to obtain the most
accurate reading.
Also when the dipstick is removed, it should be noted
whether the oil is devoid of air bubbles or not. Oil
with air bubbles gives an indication of an air leak in
the suction lines, which can cause erractic operation
and slippage. Water in the oil imparts a milky, pink
cast to the oil and can cause spewing.
EXTERNAL OIL LEAKS
Determining source of oil leakBefore attempting to correct an oil leak, the actual
source of the leak must be determined. In many
cases, the source of the leak can be deceiving due to
“wind flow” around the engine and transmission.
The suspected area should be wiped clear of all oil
before inspecting for the source of the leak. Red dyeAUTOMATIC TRANSMISSION 7C- 81
is used in the transmission oil at the assembly plant
and will indicate if the oil leak is from the transmis-
sion.The use of a “Black Light” to locate the point at
which the oil is leaking is helpful. Comparing the oil
from the leak to that on the engine or transmission
dipstick, when viewed by Black Light, will determine
the source of the leak-engine or transmission.
Oil leaks around the engine and transmission are
generally carried toward the rear of the car by air
stream. For example, a transmission oil filler tube to
case leak will sometimes appear as a leak at the rear
of the transmission. In determining the source of a
leak, proceed as follows:
1. Degrease underside of transmission.
2. Road test to get unit at operating temperature.
3. Inspect for leak with engine running.
4. With engine off, check for oil leaks due to the
raised oil level caused by drain back.
Case Porosity RepairOpel Three-Speed Automatic Transmission external
oil leaks caused by case porosity can be successfully
repaired with the transmission in the car by using the
following recommended procedures:
1. Road test and bring the transmission to operating
temperature, approximately 180 degrees F.
2. Raise car on a hoist or jack stand, engine running,
and locate source of oil leak. Check for oil leaks in
low, drive, and reverse.
3. Shut engine off and thoroughly clean area to be
repaired with a suitable cleaning solvent and a
brush- air dry. A clean, dry soldering acid brush can
be used to clean the area and also to apply the epoxy
cement.
4. Using instructions of the manufacturer, mix a suf-
ficient amount of epoxy, BUICK Group 0.423, Part
No. 1360016, or equivalent, to make the repair. Ob-
serve cautions of manufacturer in handling.
5. While the transmission case is still HOT, apply the
epoxy to the area to be repaired. Make certain the
area to be repaired is fully covered.
6. Allow cement to cure for 3 hours before starting
engine.
7. Road test and check for leaks.
Page 495 of 625
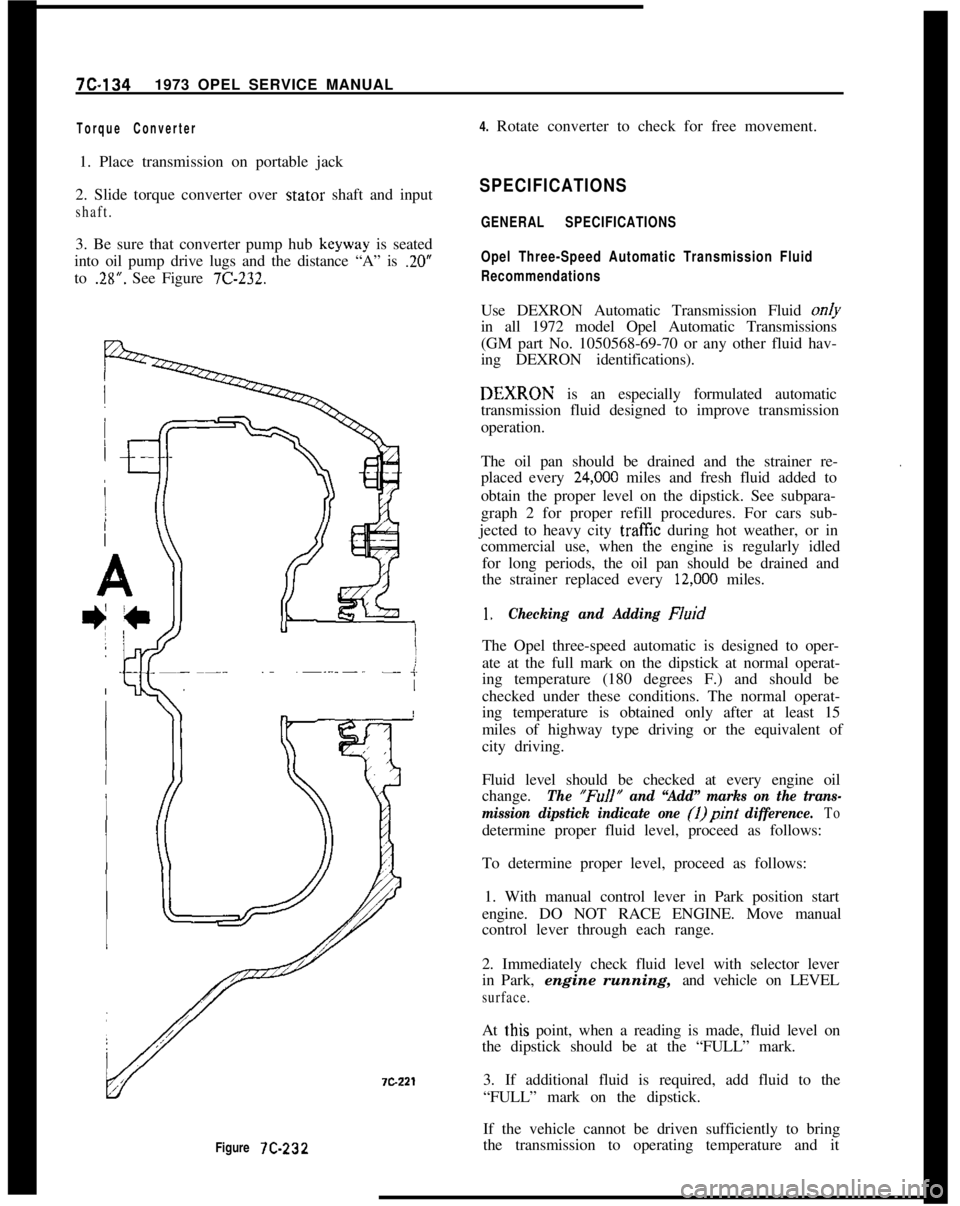
7C-1341973 OPEL SERVICE MANUAL
Figure 7C-232
Torque Converter4. Rotate converter to check for free movement.
1. Place transmission on portable jack
2. Slide torque converter over stator shaft and input
shaft.3. Be sure that converter pump hub keyway is seated
into oil pump drive lugs and the distance “A” is
.20”to
.28”. See Figure 7C-232.
SPECIFICATIONS
GENERAL SPECIFICATIONS
Opel Three-Speed Automatic Transmission Fluid
RecommendationsUse DEXRON Automatic Transmission Fluid on/y
in all 1972 model Opel Automatic Transmissions
(GM part No. 1050568-69-70 or any other fluid hav-
ing DEXRON identifications).DEXIRON is an especially formulated automatic
transmission fluid designed to improve transmission
operation.
The oil pan should be drained and the strainer re-
placed every
24,ooO miles and fresh fluid added to
obtain the proper level on the dipstick. See subpara-
graph 2 for proper refill procedures. For cars sub-
jected to heavy city
traff%z during hot weather, or in
commercial use, when the engine is regularly idled
for long periods, the oil pan should be drained and
the strainer replaced every
12,ooO miles.
.
1.Checking and Adding FluidThe Opel three-speed automatic is designed to oper-
ate at the full mark on the dipstick at normal operat-
ing temperature (180 degrees F.) and should be
checked under these conditions. The normal operat-
ing temperature is obtained only after at least 15
miles of highway type driving or the equivalent of
city driving.
Fluid level should be checked at every engine oil
change.
The “FuIl” and “Add” marks on the trans-
mission dipstick indicate one (1)pint
difference. Todetermine proper fluid level, proceed as follows:
To determine proper level, proceed as follows:
1. With manual control lever in Park position start
engine. DO NOT RACE ENGINE. Move manual
control lever through each range.
2. Immediately check fluid level with selector lever
in Park, engine running, and vehicle on LEVEL
surface.At
t,his point, when a reading is made, fluid level on
the dipstick should be at the “FULL” mark.
3. If additional fluid is required, add fluid to the
“FULL” mark on the dipstick.
If the vehicle cannot be driven sufficiently to bring
the transmission to operating temperature and it
Page 496 of 625

AUTOMATIC TRANSMISSION 7C-135
becomes necessary to check the fluid level, the trans-
mission may be checked at room temperature (70
degrees F.) as follows:
1. With manual control lever in Park position start
engine. DO NOT RACE ENGINE. Move manual
control lever through each range.
2. lmmediately check fluid level with selector lever
in Park, engine running, and vehicle on LEVEL sur-
face.At this point, when a reading is made, fluid level on
the dipstick should be I/4” below the “ADD” mark.
3. If additional fluid is required add fluid to bring
level to
l/4” below the “ADD” mark on the dip-
stick.If transmission fluid level is correctly established at
70 degrees F. it will appear at the “FULL” mark on
the dipstick when the transmission reaches normal
operating temperature (180 degrees F.) The fluid
level is set
l/4” below the “ADD” mark on the
dipstick to allow for expansion of the fluid which
occurs as transmission temperatures rise to normal
operating temperature of 180 degrees F.
Do not overfill, as foaming and loss of fluid through
the vent pipe might occur as fluid heats up. If fluid
is too low especially when cold, complete loss
of’drive may result which can cause transmission fail-
ure.
2.Draining oilpan and rep/a&g strainer assembly.
(a) Raise car on hoist or p/ace OnJxk stands, and
provide container to collect draining fluid.
(b) Remove oil pan and gasket. Discard gasket.
(c) Drain fluid from oil pan. Clean pan with solvent
and dry thoroughly with clean compressed air.
(d) Remove strainer assembly, strainer gasket and
discard.
(e) Install new oil strainer gasket. Install new strainer
assembly.
(f) Install new gasket on oil pan and install pan.
Tighten attaching bolts to 7-10 lb. ft.
(g) Lower car and add approximately three (3) pints
of transmission fluid through filler tube.
(h) With manual control lever in Park position, start
engine. DO NOT RACE ENGINE. Move manual
control lever through each range.
(i) Immediately check fluid level with selector leverin Park, engine running, and vehicle on LEVEL
sur-
face.(i) Add additional fluid to bring level to
l/4” below
the “ADD” mark on the dipstick. Do not overfill.
3.Adding Fluid to Fill Dry Transmission and Con-
verter Assembly
The fluid capacity of the Opel Three Speed Auto-
matic transmission and converter assembly is ap-
proximately IO-l/2 pints, but correct level is
determined by the mark on the dipstick rather than
by amount added. In cases of transmission overhaul,
when a complete fill is required, including a new
converter proceed as follows:
(a) Add approximately 10-l/2 pints of transmission
fluid through tiller tube.
The converter should be replaced on any major fail-
ure, such as a clutch or gearset, and an excessive
amount of foreign material is indicated in the pan. If
installation of a new converter is not required add
approximately five (5) pints of transmission fluid.
(b) With manual control lever in Park position start
engine and run at 1000 RPM. DO NOT RACE EN-
GINE. Move manual control lever through each
range.
(c) Immediately check fluid level with selector lever
in Park, engine running, and vehicle on LEVEL
sur-
face.(d) Add additional fluid to bring level to
l/4” below
the “ADD” mark on the dipstick. Do not overfill.
Opel Three Speed Automatic Transmission Towing
Instructions
If an Opel equipped with an automatic transmission
must be towed, the following precautions must be
observed:
The car may be towed safely on its rear wheels with
the shift lever in neutral position at speeds of 35 miles
per hour or less under most conditions.
However, the drive shaft must be disconnected or the
car towed on its front wheels if:
a. Tow speeds in excess of 35 mph are necessary.
b. Car must be towed for extended distances (over 50
miles).
c. Transmission is not operating properly.
If car is towed on its front wheels, the steering wheel
Page 527 of 625

98.18 1973 OPEL SERVICE MANUAL
DESCRIPTION AND OPERATION
FUNDAMENTAL PRINCIPLES OF REFRIGERATION
We all know what air conditioning does for us, but
very few understand how or why it works. An air
conditioner is functionally very similar to a refrigera-
tor, so let’s take a look at refrigeration. A refrigerator
is a simple mechanism which, surprisingly enough,
works quite a bit like a tea-kettle boiling on a stove.
That may sound far-fetched, but there is more
similarity between the two than most of us would
suspect. In fact, a modern refrigerator can make ice-
cubes and keep food cool and fresh only because a
liquid called the refrigerant boils inside the freezer.
Of codrse everyone knows a boiling tea-kettle is
“hot” and a refrigerator is “cold”. However, this is
where most of us are apt to get confused. We usually
think of “cold” as a definite, positive condition. Ac-
tually though, there is no such thing as “cold”. The
only way we can define it is in a rather negative sort
of way by saying “cold” is simply the lack of heat
just as darkness is the lack of light. We can:t make
things cold directly. All we can do is remove some
of the heat they contain and they will become cold
as a result. And that is the main job of any ice-box
or refrigerator. Both are simply devices for removing
heat.
All substances contain some heat. Theoretically, the
lowest temperature that any substance could obtain
is 459 degrees Fahrenheit below Zero. This may be
called “Cold”, and anything warmer than this con-
tains heat. Since man has never succeeded in getting
all the heat out of an object, we must think about the
transfer of heat from one object to another when
talking about controlling temperatures.
Figure
96-1 Transfer of Heat
Transfer of HeatThe only thing that will attract heat is a colder ob-ject.
:Like water, which always flows down-hill, heat
always flows down a temperature scale
- from a
warm level down to a colder one. When we hold our
hands out toward the fireplace, heat flows from the
hot fire out to our cold hands (Fig.
9B-1). When we
make a snowball, heat always flows from our warm
hands to the colder snow. In an ice-box, the ice al-
ways is colder than the stored food, so heat naturally
is drawn out of the warm food by the colder ice.
Measurement of HeatEveryone thinks he knows how heat is measured.
Thermometers are used in most: homes. Whenever
we speak of temperature from now on, we will mean
Fahrenheit. They can tell how hot a substance is, but
they can’t tell us everything about heat.
Figure
98-2 Applied Temperature Alone is Not the
Sole Measurement of Heat
When we put a tea-kettle on a stove, we expect it to
get hotter and hotter until it finally boils. All during
the process, we can tell exactly how hot the water is
by means of a thermometer (Fig.
9B-2). However,
our thermometer will show us that the flame is just
as hot when we first put the tea-kettle on the stove
as it is when the water finally boils. Why doesn’t the
water boil immediately then? Also, why does it take
longer to boil a quart of water than a cupful? Obvi-
ously temperature isn’t the only measurement of
heat.
Even though heat is intangible, it can be measured by
quantity as well as intensity. It is recognized that
thermometers indicate only the intensity of heat. The
unit for measuring quantity of heat is specified as
that amount necessary to make 1 pound of water 1
degree warmer (Fig.
9B-3). We call this quantity of
heat a British Thermal Unit. Often it is abbreviated
to Btu.
Perhaps we can get a better idea of these two charac-
Page 529 of 625
98-20 1973 OPEL SERVICE MANUAL
If we were to put a thermometer in the cold drain
water, we would see the temperature gradually creep
upwards. That is to be expected because heat is flow-
ing into the cold water making it warmer. Before
long the water would be as warm as the stored foods.
Then the water could no longer attract heat because
heat will not flow from one warm object to another
equally warm object. Since we no longer can draw
heat out of the foods we no longer are cooling them.
Now, let’s see what happens when we put ice instead
of cold water into the ice-box. This time, we’ll set the
thermometer on top of the ice (Fig. 9B-5). When wefirst look at the thermometer, it reads 32 degrees. A
couple of hours later, we open the ice compartment
door. The ice block is smaller because some of the ice
has already melted away
- but the thermometer still
reads 32 degrees. Again, still later, even more of the
ice has melted, yet the termometer continues to read
32 degrees. So long as any ice remains, no matter
how much of it has melted away, the temperature of
the ice stays right at 32 degrees.
All this time the ice has been soaking up heat, yet it
never gets any warmer no matter how much heat it
draws from the stored food. On the other hand, the
cold drain water got progressively warmer as it
soaked up heat. Why is it the addition of heat will
make water warmer yet won’t raise the temperature
of ice above the 32 degrees mark? If we till one
drinking glass with ice and another with cold water,
and put both glasses in the same room where they
could absorb equal amounts of heat from the room
air, we will find it takes much, much longer for the
ice to melt and reach room temperature than it did
for the water in the other glass to reach the same
temperature. Obviously, most of the heat was being
used to melt the ice. But it was the heat that appar-
ently disappeared or went into hiding because if
couldn’t be located with a thermometer. To best de-
scribe this disappearing heat, scientists turned to
Latin for the right word. They chose the word “la-
tent” which means hidden.
Latent Heat
So latent heat is nothing more nor less than hidden
heat which can’t be found with a thermometer.
What happens to the latent heat? Where does it
disappear to? At first it was thought it was in the
water that melted from the ice. But that wasn’t ex-
actly the right answer because, upon checking water
temperature as it melts from ice, it will be found that
it is only a shade warmer than the ice itself. It is not
nearly warm enough to account for all the heat the
ice had absorbed. The only possible answer is that
the latent heat had been used up to change the ice
from a solid into a liquid.
Many substances can be either a solid, or a liquid, ora gas. It just depends on the temperature whether
water for example was a liquid, or a solid (ice), or gas
(steam) (Fig.
9B-6).Figure 99-6 Temperature Determines State of Water
If we put some water in a tea-kettle, set it over a tire
and watch the thermometer as the water gets hotter
and hotter, the mercury will keep rising until the
water starts to boil. Then the mercury seems to stick
at the 212 degrees mark. If we put more wood on the
fire, despite all the increased heat, the mercury will
not budge above the 212 degree mark (Fig.
9B-7).Figure 98.7 Boiling Water Never Exceeds 2 12
DegreesEven though many housewives won’t believe it, no
matter how large or hot you make the flame, you
can’t make water hotter than 2 12 degrees. As a liquid
changes into a gas, it absorbs abnormally great
amounts of heat without getting any hotter. Here is
another instance where heat disappears.
Now we have two different kinds of latent heat,
which are quite alike. To keep their identities sepa-
rate, the first one is called latent heat of fusion. Since
fusion means the same as melting, it is a good de-
scriptive name. The other kind is called latent heat
of vaporization because‘ that means the same as
evaporation.
It may seem as though we have drifted into a story
Page 530 of 625
REFRIGERANT COMPONENTS ALL MODELSSE- 21
about heat instead of refrigeration. But in doing so,
we have learned how a simple ice-box works. It’s
because the magic of latent heat of fusion gives ice
the ability to soak up quantities of heat without get-
ting any warmer.
Therefore, since it stays cold, it can continue to draw
heat away from stored foods and make them cooler.
The latent heat of vaporization can be an even better
“magnet” because it will soak up even more heat.
Whenever we think of anything boiling, we instinc-
tively think of it being very hot. However, that’s not
true in every case. Just because water
boi1.s at 212
degrees doesn’t mean that all other substances will
boil at the same temperature. Some would have to be
put into a blast furnace to make them bubble and
give off vapor. On the other hand, others will boil
violently while sitting on a block of ice.
And so each substance has its own particular boiling
point temperature. But regardless of whether it is
high or low, they all absorb unusually large quanti-
ties of heat without getting any warmer when they
change from a liquid into a vapor.
Consequently, any liquid that will boil at a tempera-
ture below the freezing point of water, will make ice
cubes and keep vegetables cool in a mechanical re-
frigerator.
Figure
9B-10 Simple R-12 Refrigerator
Refrigerant - 12Refrigerant-12 is used in the air conditioning system
and boils at 21.7 degrees below zero. Maybe that
doesn’t mean very much until we picture a flask of
R-12 sitting at the North Pole boiling away just like
a tea-kettle on a stove. No one would dare pick up
the flask with his bare hands because, even though
boiling, it would be so cold and it would be drawing
heat away from nearby objects so fast that human
flesh would freeze in a very short time. If we were toput a flask of R-12 inside a refrigerator cabinet, it
would boil and draw heat away from everything sur-
rounding it (Fig.
9B-10). So long as any refrigerant
remained in the flask, it would keep on soaking up
heat until the temperature got down to 21.7 degrees
below zero.
Now we can begin to see the similarity between a
boiling tea-kettle and a refrigerator. Ordinarily we
think of the flame pushing heat into the tea-kettle.
Yet, it is just as logical to turn our thinking around
and picture the tea-kettle pulling heat out of the
flame. Both the tea-kettle and the flask of refrigerant
do the same thing they draw in heat to boil
although they do so at different temperature levels.
There also is another similarity between the ice-box
and the mechanical refrigerator. In the ice-box, wa-
ter from melting ice literally carried heat out of the
cabinet. In our simple refrigerator, rising vapors do
the same job.Rdsing
Our R-l 2Water is so cheap that we could afford to throw it
away. But R-12, or any other refrigerant, is too ex-
pensive just to let float away into the atmosphere. If
there was some way to remove the heat from the
vapor and change it back into a liquid, it could be
returned to the flask and used over again (Fig. 9B-
11).There is a way, and that is where we find the biggest
difference between the old ice-box and the modern
refrigerator. We used to put in new ice to replace that
lost by melting. Now we use the same refrigerantover and over again.
Figure 9B-1 1 Re-Using Refrigerant
Page 531 of 625
9B-22 1973 OPEL SERVICE MANUAL
We can change a vapor back into a liquid by chilling
it, or do the same thing with pressure. When we
condense a vapor we will find that the heat removed
just exactly equals the amount of heat that was neces-
sary to make the substance vaporize in the first place.
At last the lost is found! The latent heat of vaporiza-
tion the heat that apparently disappeared when
a liquid boiled into a vapor again reappears on
the scene when that same vapor reverts back into a
liquid. It is just like putting air into a balloon to
expand it and then letting the same amount of air out
again to return the balloon to its original condition.
We know that any substance will condense at the
same temperature at which it boiled. This tempera-
ture point is a clear-cut division like a fence. On one
side, a substance is a liquid. Immediately on the
other side it is a vapor. Whichever way a substance
would go, from hot to cold or cold to hot, it will
change its character the moment it crosses over thefence.But pressure moves the fence! Water will boil at 212
degrees under normal conditions. Naturally, we ex-
pect steam to condense at the same temperature. But
whenever we put pressure on steam, it doesn’t! It will
condense at some temperature higher than 212 de-
grees. The greater the pressure, the higher the boiling
point and the temperature at which a vapor will
condense. This is the reason why pressure cookers
cook food faster, since the pressure on the water
permits it to boil out at a higher temperature. We
know that R-12 boils at 21.7 degrees below zero. A
thermometer will show us that the rising vapors,
even though they have soaked up lots of heat, are
only slightly warmer. But the vapors must be made
warmer than the room air if we expect heat to flow
out of them. Also, the condensing point temperature
must be above that of room air or else the vapors
won’t condense.This is where pressure comes to the rescue. With
pressure, we can compress the vapor, thereby con-
centrating the heat it contains. When we concentrate
heat in a vapor that way, we increase the intensity of
the heat or, in other words, we increase the tempera-ture;because temperature is merely a measurement
of heat intensity. And the most amazing part of it all
is that we’ve made the vapor hotter without actually
adding any additional quantity of heat (Fig.
9B-12).
Use of Pressure in RefrigerationBecause we must live by press&s and gauges in air
conditioning work, the following points are men-
tioned so that we will all be talking about the same
thing when we speak of pressures.
All pressure, regardless of how it is produced, is
measured in pounds per square inch (psi).Figure 98.12 Compressing a Vapor Concentrates its
HeatAtmospheric Pressure is pressure exerted in every
direction by the weight of the atmosphere. At higher
altitudes air is raritied and has less weight. At sea
level atmospheric pressure is 14.7 psi.
Any pressure less than atmospheric is known as a
partial vacuum or commonly called a vacuum. A
perfect vacuum or region of no pressure has never
been mechanically produced. Gauge pressure is used
in refrigeration work. Gauges are calibrated in
pounds (psi) of pressure and inches of Mercury for
vacuum. At sea level
“0” lbs. gauge pressure is
equivalent to 14.7 lbs. atmospheric pressure. Pres-
sure greater than atmospheric is measured in pounds
(psi) and pressure below atmospheric is measured in
inches of vacuum. The “0” on the gauge will always
correspond to the surrounding atmospheric pressure,
regardless of the elevation where the gauge is being
used.
Basic Refrigerator OperationWe’ve now covered all the ground-rules that apply to
refrigeration. Most likely they still are a little hazy,
but it is easy enough to remember these main points.
All liquids soak up lots of heat without getting any
warmer when they boil into a vapor, and, we can use
pressure to make the vapor condense back into a
liquid so it can be used over again. With just that
amount of knowledge, here is how we can build a
refrigerator.
We can place a flask of refrigerant in an ice-box. We
know it will boil at a very cold temperature and will
draw heat away from everything inside the cabinet
(Fig. 9B-13).
We can pipe the rising vapors outside the cabinet and
thus provide a way for carrying the heat out. Once
Page 532 of 625

REFRIGERANT COMPONENTS ALL MODELS96.23Figure 96-l 3 Basic Refrigerant Circuit
we get the heat-laden vapor outside, we can com-
press it with a pump. With enough pressure, we can
squeeze the heat out of “cold” vapor even in a warm
room. An ordinary.radiator will help us get rid of
heat.
By removing the heat, and making the refrigerant
into a liquid, it becomes the same as it was before, So,
we can run another pipe back into the cabinet and
return the refrigerant to the flask to be used over
again.
That is the way most mechanical refrigerators work
today. Now, let’s look at an air conditioning unit to
see how closely it resembles the refrigerator we have
just described.
Basic Air ConditionerWhen we look at an air conditioning unit, we will
always find a set of coils or a tinned radiator core
through which the air to be cooled passes. This is
known as the “evaporator” (Fig.
9B-14). It does the
same job as the flask of refrigerant we
spok.e about
earlier. The refrigerant boils in the evaporator. In
boiling, of course, the refrigerant absorbs heat and
changes into a vapor. By piping this vapor outside
the car we can bodily carry out the heat that caused
its creation.
Once we get vapor out of the evaporator, all we haveFigure 98.14 Evaporator Assembly
to do is remove the heat it contains. Since heat is the
only thing that expanded the refrigerant from a liq-
uid to a vapor in the first place, removal of that same
heat will let the vapor condense into a liquid again.
Then we can return the liquid refrigerant to the
evaporator to be used over again.
Actually, the vapor coming out of the evaporator is
very cold. We know the liquid refrigerant boils at
temperatures considerably below freezing and that
the vapors arising from it are only a shade warmer
even though they do contain quantities of heat.
Consequently, we can’t expect to remove heat from
sub- freezing vapors by “cooling” them in air tem-
peratures that usually range between 60 and 100
degrees heat refuses to
flow from a cold object
toward a warmer object.
But with a pump, we can squeeze the heat-laden
vapor into a smaller space. And, when we compress
the vapor, we also concentrate the heat it contains.
In this way, we can make the vapor hotter without
adding any heat. Then we can cool it in compara-
tively warm air.
That is the only responsibility of a compressor in an
air conditioning system (Fig.
9B-15). It is not in-
tended to be a pump just for circulating the refriger-
ant. Rather, its job is to exert pressure for two
reasons. Pressure makes the vapor hot enough to
cool off in warm air. At the same time, the compres-
sor raises the refrigerant’s pressure above the con-
densing point at the temperature of the surrounding
air so it will condense.
As the refrigerant leaves the compressor, it is still a
vapor although it is now quite hot and ready to give
up the heat that is absorbed in the evaporator. One
of the easiest ways to help refrigerant vapor dis-
charge its heat is to send it through a radiator- like
contrivance known as a condenser (Fig. 9B-16).
The condenser really is a very simple device having
no moving parts. It does exactly the same job as the
radiator in a typical steam-heating system. There,
the steam is nothing more than water vapor. In pass-
ing through the radiator, the steam gives up its heat
and condenses back into water.
The same action takes place in an air conditioning
Page 535 of 625

98-26 1973 OPEL SERVICE MANUAL
greater than the opposing pressure in the power ele-
ment. Therefore, the valve remains closed. When the
compressor is started, it will reduce the pressure and
temperature of the refrigerant in the cooling coil to
a point where the vapor pressure in the power ele-
ment becomes the stronger. The seat then moves off
the orifice and liquid starts to flow through the valve
orifice into the cooling coil.
The purpose of the power element is to help deter-
mine the quantity of liquid that is being metered into
the cooling coil. As the temperature of the low pres-
sure line changes at the bulb, the pressure of
the
vapor in the power element changes, resulting in a
change of the position of the seat. For example, if the
cooling coil gets more liquid than is required, the
temperature of the low pressure line is reduced and
the resultant lowering of the bulb temperature
reduces the pressure of the vapor in the power ele-
ment, allowing the seat to move closer to the orifice.
This immediately reduces the amount of liquid leav-
ing the valve. Under normal operation, the power
element provides accurate control of the quantity of
refrigerant to the cooling coil.
To employ our tire pump analogy once more for
clarity, it is the same situation that would exist if you were inflating a tire with a very slow leak. Providing
you pumped the air into the tire as fast as it leaked
out, you would be able to maintain pressure even
though the air would merely be circulating through the tire and leaking out through the puncture.
To Sum Up
So far, we’ve discussed only what each unit in an air
conditioning system does. We’ve learned that the
evaporator is the unit in which liquid refrigerant
soaks up heat from the air, the compressor is a pump
for squeezing this heat out of the vapor, the con-
denser is a radiator for getting rid of the heat, and the
thermostatic expansion valve is a device for regulat-
ing the pressure on the refrigerant. Now, let’s
find
out how the temperature of the cooled air is con-
trolled.
METHOD OF TEMPERATURE CONTROL
To achieve temperature control, the compressor is
run intermittently, automatically turning on and off
as necessary to maintain proper temperature.
Thermostatic Switch
The compressor can be started and stopped au-
tomatically through the use of an electro-magnetic
clutch and a thermostat affected by variations of temperature.
The job is usually done by a gas bulb thermostat (Fig.
9B-21).
Figure 9B-21 Thermostatic Switch Schematic
With the gas bulb type of thermostat, a highly expan-
sive gas is sealed into a metallic bulb which is located
in the air stream as it leaves the evaporator. A small
tube leads from the bulb to a bellows operated switch. As air temperature rises, the gas inside the
bulb expands, travels through the tube to the bellows
and closes the electrical switch that engages the com-
pressor clutch.
Of course, as soon as the compressor starts running,
the temperature begins to go down. As the air being
cooled gets colder, the gas in the thermostat bulb
begins to reduce the pressure on the switch bellows.
This
Ilips “off’ the switch and disengages the com-
pressor clutch.
REFRIGERANTS
No matter how scientifically refrigerating machinery
is built or how
efftciently it runs, it alone cannot
remove heat. The only thing that carries heat out of
a refrigerator cabinet or an automobile is the sub-
stance we call the refrigerant.
There are many refrigerants known to man. In fact,
any liquid that can boil at temperatures somewhere
near the freezing point of water can be used.
But a boiling point below the temperature at which
ice forms is not the only thing that makes a good
refrigerant. A refrigerant should also be non-
poiso-
nowand non-explosive to be safe. Besides that, we
want a refrigerant that is non-corrosive and one that
will mix with oil.
Since Nature did not provide an ideal refrigerant,
chemists went to work to see if they could do any
better. They did! But it wasn’t as simple as that.
At first, they tried to improve existing natural refrig-
erants. But after exploring innumerable trails along
Page 537 of 625
9B-28 1973 OPEL SERVICE MANUAL
Thus, from the standpoint of comfort, complete air
conditioning should control the relative humidity of
the air as well as its temperature.
By reducing the humidity, we sometimes can be just
as “cool” in a higher room temperature than other-
wise would be comfortable. Laboratory tests have
shown that the average person will feel just as cool
in a temperature of 79 degrees when the relative
humidity is down around 30 percent as he will in a
cooler temperature of 72 degrees with a high relative
humidity of 90 percent.
There are practical limits though within which wemust stay when it comes to juggling humidity. For
human comfort, we can’t go much below a relative
humidity of 30 percent because anything lower than
that would cause an unpleasant and unhealthy dry-
ness in the throat and nasal passages.
Summertime temperatures of 85 degrees sometimes
bring with them relative humidities around 75 to 80
percent. Some coastal cities have relative humidities
averaging as high as 87 percent. To gain maximum
human comfort, an air conditioning system should
cool the air down and reduce the humidity to com-
fortable limits.
The cooling job usually is done just as it is in a
refrigerator. A compressor sends refrigerant through
a chilling unit where it absorbs heat. The heat is
drawn out of the air which circulates through the
chilling unit. Along with the cooling job it does, the
evaporator unit also removes much of the moisture
from the air. Everyone is familiar with the sight of
thick frost on the freezer of a refrigerator. That frost
is simply frozen moisture that has come out of the
air.
Figure 99.22 Condensation
The evaporator unit in an air-conditioning system
does the same thing with this one exception. Becauseits temperature is above the freezing point, the mois-
ture does not collect in the form of ice or frost.
Instead, the moisture remains fluid and drips off the
chilling unit. This action is similar to what occurs on
the cool bathroom mirror when a hot shower is
turned on (Fig. 9B-22). A further advantage of airconditioning is that dust and pollen particles are
trapped by the wet surfaces of
.the evaporator core
and then drained off with the condensed moisture.
This provides very clean, pure air for breathing, and
is of great benefit to those who suffer from asthma
or ahergies such as hay fever.
Basic Refrigeration CycleLet’s review the basic refrigeration cycle. Keep this
basic cycle in mind because knowledge of the cycle,
knowledge of the particular system you are working
on and proper use of the gauges will permit quick,
accurate diagnosis of problems as they arise.
Any refrigeration system takes advantage of the
principles just described. The air conditioning sys-
tem illustrated in Fig. 9B-23 contains
five basic parts;
a compressor, a condenser, a receiver, an expansion
valve and an evaporator. Assuming R-12 as our re-
frigerant, let us follow through the refrigeration cy-
cle.Refrigerant gas under low pressure is drawn into the
compressor where it is compressed to a high pres-
sure. During compression, the refrigerant gas is
heated. When sufficient pressure is built up, the hot
gas passes into the condenser where it cools by giving
off heat to the air passing over the condenser sur-
faces.As the refrigerant gas cools, it condenses into a liquid
at high pressure and accumulates in the receiver. The
high pressure liquid refrigerant passes to the expan-
sion valve at the entrance to the evaporator. At the
valve orifice the pressure is lowered and the refriger-
ant enters the evaporator core as a low pressure liq-
uid. When the refrigerant is exposed to the lower
evaporator pressure, it begins to boil and is changed
to a vapor state. As the refrigerant passes through
the evaporator, it continues to boil by absorbing heat
from the air passing over the evaporator surfaces
until it is completely vaporized. From the evaporator
the cool low pressure refrigerant gas is drawn back
to the compressor and the cycle repeated.
Thus the air passing over the evaporator surfaces is
cooled simply by giving up heat to the refrigerant
during the boiling process.
CHEMICAL INSTABILITY AND REFRIGERATING
SYSTEM FAILURESA sealed refrigerating system is a complex physical-
chemical combination which is designed for stability